 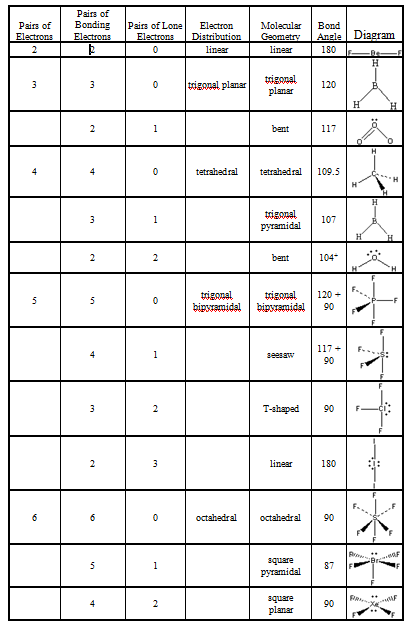
It means the molecule will produce four new hybrid orbitals of equal energy which corresponds to sp3 hybridization.Īs we know in a single covalent bond, the formation of only sigma (σ) bonds takes place with no pi (π) bonds. The steric number is equal to the lone pairs and sigma bonds the central atom has.Īs a single phosphorus trifluoride (PF3) molecule has three bonds (between phosphorus and fluorine) and one lone pair of electrons, the steric number is four. To figure out the hybridization of the central atom, it is essential to determine the steric number in the phosphorus trifluoride (PF3) molecule. Hybridization is a method of combining atomic orbitals of the same atom to produce new orbitals which are called hybrid orbitals. Hybridization in Phosphorus Trifluoride (PF3) molecule You can also check an article related to the polarity of PF3. This repulsion distorts the whole structure where the effect increases to many folds because of the smaller size of fluorine and shorter atomic radius distance.ĭue to its original pyramidal shape, the PF3 molecule turns out to be polar. When any shared pair of electrons come in the near vicinity of lone pair of electrons, as do not want to bond. Because they are highly stable, their repulsion is stronger than that of the bonding pair of electrons. There exist a lone pair of electrons on the phosphorus which does not participate in the bond formation. As the lone pair repulsion is stronger than the bond pair or bond pair-lone pair repulsion, it reduces the bond angle. This anomaly is due to the lone pair of electrons, and the smaller size of the fluorine atom. This angle makes the structure bent where the ideal bond angle for the bent, trigonal pyramidal structure is 109.5°. This theory explains that the bond angle between the fluorine-phosphorus-fluorine (F-P-F) is 97°. The geometrical structure of the tetra-atomic Phosphorus Trifluoride (PF3) molecule is studied with the help of the Valence Shell Electron Pair Repulsion (VSEPR) theory. Geometrical Structure of Phosphorus Trifluoride (PF3) molecule Find the central atom to draw the Lewis structure, which is phosphorus in the case of phosphorus trifluoride (PF3). It is three covalent single bonds, each between phosphorus and fluorine atom with the presence of no double or triple bonds.Ĥ. Find the total number of bonds forming in a single PF3 molecule. It is six in total, where three valence electrons are needed by the phosphorus atom and one, each by three fluorine atoms.ģ. Find out the number of valence electrons further needed of a single PF3 molecule to stabilize itself. Find out the total number of valence electrons in PF3, which is 26.Ģ. PF3 is a tetra-atomic molecule where phosphorus donates three valence electrons, and three fluorine atoms accept one electron each to undergo a bond formation and reach a stable condition.īelow are the steps to draw the lewis structure of the PF3 moleculeġ. 
The Lewis structure is drawn using eight dots of valence electrons around the symbols of the atom with lines showing bond formation. Lewis structure of Phosphorus Trifluoride (PF3)  
Whereas, on the other hand, the atomic number of fluorine is nine which makes its electronic configuration 1s2 2s2 2p5, having a scarcity of only one valence electron. As we know, the p shell can hold a maximum of six electrons there is a scarcity of three electrons. The atomic number of phosphorus is fifteen which makes its electronic configuration 1s2 2s2 2p6 3s2 3p3. The reason for the same is explained with the help of their electronic configuration. So, fluorine accepts the electron whereas, the phosphorus atom tends to donate the electrons to complete their octet and reach a stable condition. Here, it is essential to understand that a higher number of valence electrons will strengthen the ability of an atom to accept the electrons rather than donating. Moreover, uneven or unpaired electrons compel them to participate in the bond formation. Because they are present in the outermost shell, the hold of the nucleus is weak on them. The electrons present in the outermost shell of an atom are called valence electrons. Valence electrons in Phosphorus and Fluorine atom On the other hand, a single fluorine atom has seven valence electrons with a d of one to complete its octet and reach a stable condition. One phosphorus atom has five valence electrons, having a scarcity of three to complete its outermost shell or octet. As per this rule, the maximum number of valence electrons an atom can have is eight. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
